Most of the energy released in fission process is in process of _Ĭlarification: Most of the energy released is in the form of kinetic energy and is absorbed by fission products. It is derived from Greek word ‘isos’ meaning EQUAL and ‘baros’ meaning WEIGHT.ħ. The term isobar was suggested by Alfred Walter Stewart in 1918. Atoms of different chemical elements that have the same number of nucleons are called as?Ĭlarification: Atoms of different chemical elements that have the same number of nucleons are called as isobars. 
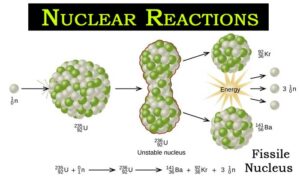
Frisch named Frisch names the process by analogy with biological fission of living cells.Ħ. This isotope is highly unstable which lasts for one millionth of a second and splits into two equal parts releasing energy of 200MeV.Ĭlarification: Nuclear fission of heavy metals was discovered by German Otto Hahn on Decemand was explained theoretically by Lise Meitner and her nephew Otto Robert Frisch on 1939. What happens when a neutron is absorbed by a nucleus of an atom of U 235?Ĭlarification: When a neutron is absorbed by a nucleus of an atom U 235, a U 236 isotope is formed. Only neutrons can result in sustained reaction as two or three neutrons are released for each one absorbed by fission.Ĥ. Thus for a sustained reaction, eve neutrons with lower energy should be capable of causing fission. In order to not waste the nuclear fuelĬlarification: Due to collisions with various nuclei, initial high kinetic energy of fission neutron decreases. Why neutrons with lower energy should be capable of causing fission?ĭ. All fission species should have long half lives so that the rate of decay is not so fast.ģ. Heavy nuclei must be such that they can be fissioned by neutrons of an energy such substance are called?Ĭlarification: Heavy nuclei must be such that they can be fissioned by neutrons of energy such substance are called Fission species. Two to three neutrons are emitted per nucleuses which are known as fission elements.Ģ. This result in decrease in mass and consequent exothermic energy and emission of neutrons take place. In which of the following process are Neutrons emitted?Ĭlarification: Nuclear fission is the process in which a heavy nucleus is split into two or more lighter nuclei. Within one year after having been unloaded from the reactor, the activity contained in the irradiated fuel decreases to about 1/100 of the original level and slowly decreases further in the following years.Energy Engineering Multiple Choice Questions on “Nuclear Fission & Nuclear Fusion”.ġ. The water largely shields the radiation and at the same time absorbs the generated residual heat. Therefore the spent fuel elements are initially stored in a water-filled pool inside the nuclear power plant ( spent fuel pool). If the heat would not be removed, this so-called residual heat would increase the temperature far beyond the melting point of the fuel elements. The fission products generating inside the fuel elements are radioactive and generate large amounts of heat, even after the reactor has been shut down. 
In the case of a pressurised water reactor, water with added boron is additionally fed into the reactor for permanent subcriticality. The control rods catch the neutrons generated in the reactor and thus end the nuclear chain reaction. 
Subcriticality is achieved by lowering the neutron-absorbing control rods between the fuel elements in the reactor core. To shut down a nuclear power plant, the reactor must be brought into a permanently uncritical state (subcriticality) and the heat that continuous to generate must be discharged safely. This chain reaction is maintained by the fact that more neutrons are produced in the fission process than are used in the absorption process. During the power operation of a nuclear power plant, a self-sustaining chain reaction occurs in the reactor core. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
